Reduced mass of two particles.
A particle-1 with mass m1 and a particle-2 with mass m2 are influencing each other by force f(r)
(Fig.1) two particles m1 and m2
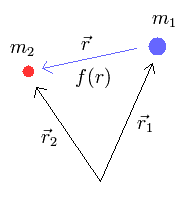
Here we derive the reduced mass μ = m1m2/(m1+m2) in a particle-1 with mass m1 and a particle-2 with mass m2 influencing each other by the force f(r).
(Eq.1)
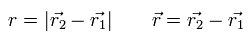
r1 and r2 means the vectors of positions of an electron-1 and an electron-2.
r means the vector from the electron-1 towards the elecrtron-2.
(Eq.2) Force = f(r)
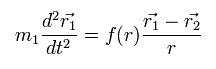
Eq.2 is the force f(r) from the particle-2 acting on the particle-1 of mass m1.
(Eq.3)
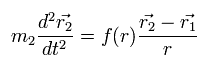
Due to action and reaction law, the particle-2 of mass m2 also feels the force f(r) with the same magnitude in the opposite direction from the particle-1.
(Eq.4)
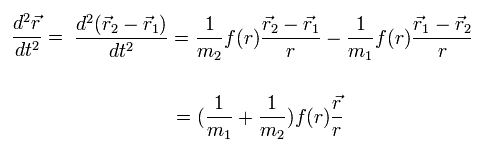
From Eq.1, Eq.2, Eq.3, we obtain Eq.4.
Reduced mass μ = m1m2/(m1 + m2)
(Eq.5) Reduced mass = μ
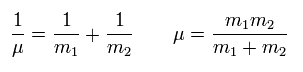
(Eq.6) Only the particle-2 of reduced mass μ appears to be moving
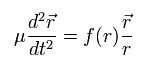
From Eq.4 and Eq.5, we can think only the particle-2 of the fictitious reduced mass μ appears to be moving around the stationary particle-1 by force f(r) between them.
We can treat only one particle-2 is moving.
When we use reduced mass μ, we can treat two moving pariticles-1 and 2 as if only one particle-2 of the reduced mass μ is moving around the particle-1 that appears to stop.
(Eq.7) R = center of the whole mass
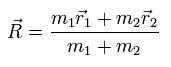
In Eq.7, R is the center of mass of the entire particle-1 and particle-2.
(Eq.8)
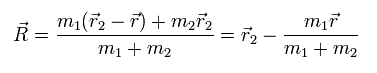
(Eq.9)
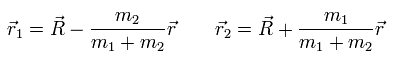
From Eq.7, we obtain Eq.8 and and Eq.9.
Only one particle of reduced mass is moving.
If we use reduced mas μ, two moving particles-1 and 2 can be treated as if only one particle of the fictitious reduced mass is moving around the other stationary particle.
(Eq.10) Only kinetic energy of only one particle with reduced mass μ needs to be considered even in two moving particles-1 and 2.
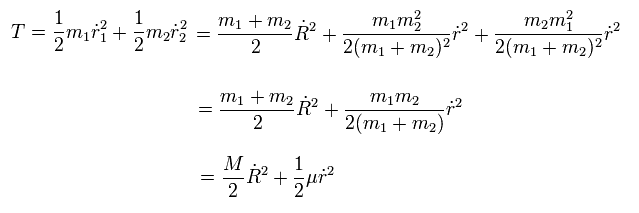
From Eq.6 ~ Eq.9, we can equate the total kinetic energy summing two moving particles-1 and 2 (= center of the entire mass R is stationary ) to the kinetic energy of only one particle of fictitious mass μ, as shown in the above Eq.10 ( this-p.2 ).
Actually, the total energy of a hydrogen atom calculated by Schrodinger equation equal to real Bohr model ( this-p.10-(4.130), this-(4.133) ) using the reduced mass μ (= almost the same as the original electron mass ) is known to agree with experimental hydrogen energy ( this-p.19 ).
↑ So an electron and a nucleus are actually moving inside an atom, which contradicts the unrealistic quantum mechanical static electron's probability electron wavefunction.
There are two points where a helium nucleus stops
The center of mass of two electrons and a helium nucleus are attracting and oscillating periodically like a two-body spring where the nucleus stops in two points (= when the angle of electron-1-nucleus-electron-2 is 90o ).
(Fig.2) When a helium nucleus is "oscillating" instead of rotating
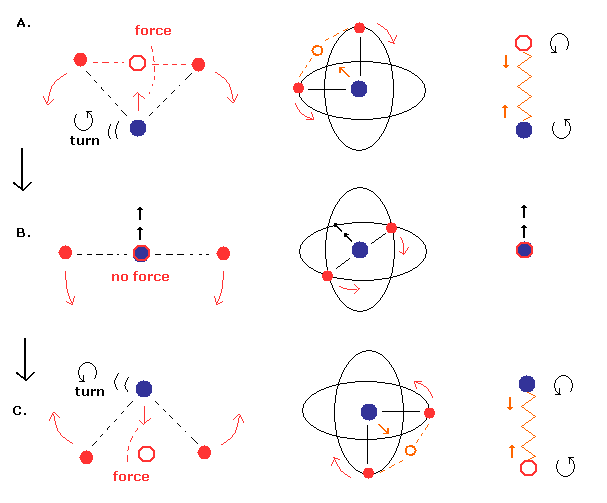
A helium nucleus is oscillating linearly.
A helium nucleus oscillating linearly attracted by the center of two electrons transiently stops only when making U-turn.
Unlike the one-electron hydrogen atom, in a helium consisting of two symmetrical electrons and a nucleus, there are at least two points where a helium nucleus transiently stops, and the reduced mass does not need to be used only in these two points.
The above Fig.2 shows the special case when the center C of two electrons (= e1 and e2 ) of total mass of 2me (= me is one electron's mass ) and the helium nucleus N are just oscillating linearly, attracting each other, like a spring.
↑ In this linear spring-like motion, there are two points where the helium nucleus N transiently stops (= where the reduced mass μ does not need to be used ) when making U-turn.
Reduced mass of a helium
The sum of two electrons satisfies the usual reduced mass of two-body system.
(Fig.3) Reduced mass of Helium atom.
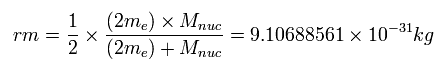
A helium electron's reduced mass (= rm )
Each electron's reduced mass (= rm ) of a helium is half of the ordinary two-body reduced mass between the whole two electrons (= 2me ) and the helium nucleus .
Except in two points when a nucleus transiently stops and makes U-turn, a helium nucleus (= N ) is moving and oscillating attracted by two electrons' center of mass (= C ).
So we can treat the center of the whole two electrons of the total mass 2me ( me is one electron's mass ) and a helium nucleus as two-body reduced mass system.
So we can treat each electron as if it has the fictitious reduced mass (= rm ), which is half the ordinary two-body reduced mass where only the helium nucleus appears to stop.
A helium nucleus stops only when an electron-1 is in y-axis (= at the start ).
An electron-1 (or electron-2 ) starts from y-axis (or z-axis ) where a helium nucleus transiently stops (= so no reduced mass ) to x-axis, moving 1/4 orbit where reduced mass is used.
(Fig.4) Electrons have moved one quarter of their orbits.

When a helium nucleus is oscillating linearly
We calculate the case when a helium nucleus is oscillating linearly, attracted by the center of two symmetrical electrons, using programs below.
sample JAVA program (= the nucleus is oscillating like Fig.2)
C language program
In these programs, as shown in the above Fig.4, the electron-1 starts from y-axis or y-coordinate of r2 toward the x-axis, moving its 1/4 orbit, while the electron-2 symmetrically starts from z-axis (= z-coordinate is r2 ) toward x-axis.
↑ A helium nucleus transiently stops only when making U-turn (= only when the electron-1 exists in y-axis at the start ) where ordinary electron mass (= me ) instead of reduced mass (= rm ) μ is used when calculating the initial electrons' kinetic energy.
Except when the electron-1 exists in y-axis at the start (= where the angle of electron-1-nucleus-electron-2 is 90 degree ), the helium nucleus is moving or oscillating linearly (= instead of rotating ) where a reduced mass must be used to make the nucleus appear to stop.
This helium model with reduced mass perfectly agrees with experiments.
The predicted helium energy (= -79.0071 eV ) whose nucleus is oscillating linearly also agrees with experimental helium energy of -79.005 eV
| Helium energy (eV) | r2 (MM) | 1/4 orbit wavelength | one-orbit wavelength |
|---|---|---|---|
| -79.0000 | 3137.0 | 0.250011 | 1.000044 |
| -79.0037 | 3136.5 | 0.250006 | 1.000024 |
| -79.0071 | 3136.5 | 0.250000 | 1.000000 |
| -79.0100 | 3136.0 | 0.249996 | 0.999984 |
| -79.0300 | 3135.5 | 0.249964 | 0.999856 |
As shown in the above table-1, the real Bohr's helium model's predicted energy (= -79.0071 eV ) whose nucleus is oscillating linearly also perfectly agrees with the experimental helium energy of -79.005 eV (= sum of 1st and 2nd ionization energies of helium ) in executing these programs.
The whole helium is usually rotating.
When the whole helium is rotating instead of linearly oscillating, a helium nucleus transiently stops (= spinning ) only when two electrons are just at the opposite side of a nucleus, which also agrees with experiments.
(Fig.5) The whole nucleus and two electrons are "rotating".
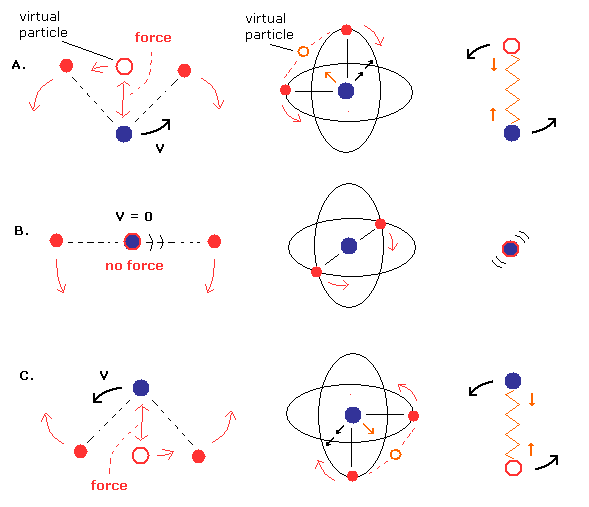
A helium nucleus is actually rotating.
A helium nucleus and the center of two electrons are rotating to approach and go away from each other where the nucleus transiently stops only when the center of two electrons is just at the nucleus.
The actual helium nucleus is usually rotating as shown in the above Fig.5 instead of oscillating linearly.
As shown in the above Fig.5, generally, the helium nucleus and the center of two symmetrical electrons attracting each other is rotating instead of oscillating linearly.
In this general helium model whose nucleus and two electrons are rotating, only when the center of two electrons are just at the helium nucleus, the nucleus appears to stop (= spinning instead of moving in one particular direction = the above Fig.5-middle due to conserving the total angular momentum ) where the normal electron mass instead of reduced mass should be used.
As shown in the real photoelectric effect without the unreal quantum mechanical virtual photons with imaginary mass, only de Broglie wave generated by the moving particles (= not spinning ) is relevant to the measured kinetic energy.
The substance of the medium transmitting light, Coulomb force and de Broglie wave is originally moving around as fast as light speed, so its spinning motion (= usually slower than light ) not generating de Broglie wave does not contribute to the kinetic energy.
Case of the whole helium is rotating
An electron-1 (or an electron-2 ) starts from x-axis where a helium nucleus transiently stops (= so only at this point, no reduced mass is used ) to y-axis, moving its 1/4 orbit with reduced mass
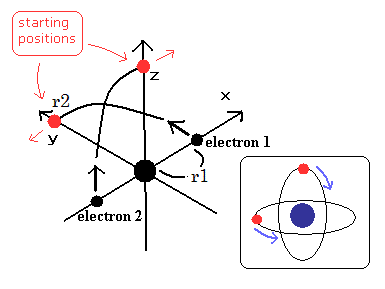
(Fig.6) Starting positions of Fig.5 model.
Real Bohr's helium agrees with experiments
Real Bohr's helium model with a rotating nucleus or reduced mass also perfectly agrees with experimental helium energy.
In the general case of a helium rotating, only when the electron-1 ( and electron-2 ) start from x-axis (= a helium nucleus is just at the center of two electrons ), the ordinary electron mass instead of reduced mass is used (= because only at this point, a helium nucleus stops, spinning, Not moving ).
As shown in the normal real helium model using this or this program, the electron-1 starts from the x-axis (= initial x-coordinate is r1 ) with initial kinetic energy that is obtained not using reduced mass toward the y-axis, moving its 1/4 orbit. ← After electrons start to move, reduced mass is used.
↑ The predicted helium energy (= -79.0037 eV ) in this normal rotating helium model also perfectly agrees with the experimental helium energy of -79.005 eV ↓.
The rotating helium agrees with experiments.
Energy of the rotating helium predicted by this Bohr model (= -79.0037 eV ) perfectly agrees with experimental helium energy of -79.005 eV
| Helium energy (eV) | r1 (MM) | 1/4-orbit wavelength | one-orbit wavelength |
|---|---|---|---|
| -78.800 | 3082.0 | 0.250323 | 1.001292 |
| -79.0000 | 3074.0 | 0.250006 | 1.000024 |
| -79.0037 | 3074.0 | 0.250000 | 1.000000 |
| -79.0100 | 3074.0 | 0.249990 | 0.999960 |
| -79.200 | 3067.0 | 0.249690 | 0.998760 |
This Bohr's helium agrees with experiments even without using reduced mass.
Energy of this real Bohr's helium model without using reduced mass (= only original electron mass is used = -79.0485 eV ) also agrees with experimental helium energy of -79.005 eV
| Helium energy (eV) | r1 (MM) | 1/4-orbit wavelength | one-orbit wavelength |
|---|---|---|---|
| -79.0037 | 3075.0 | 0.250071 | 1.000284 |
| -79.0300 | 3074.0 | 0.250030 | 1.000120 |
| -79.0485 | 3073.5 | 0.250000 | 1.000000 |
| -79.0600 | 3073.0 | 0.249982 | 0.999928 |
| -79.0800 | 3072.0 | 0.249950 | 0.999800 |
Bohr's helium without reduced mass also agrees with experiments
Even if we use the original electron mass instead of reduced mass in all calculations, Bohr's model agrees with experimental energy of -79.005 eV
Even if we use the original electron mass instead of the reduced mass in all calculations, this real Bohr's helium's predicted energy (= -79.0485 in the above table.3 ) agrees with the experimental helium energy of -79.005 eV (= sum of 1st and 2nd ionization energies of the helium ).
If you run one of the programs below, and choose the condition of "2 (= Not reduced mass)", this program calculates the helium energy Not using reduced mass (= always using the original electron mass in all computation ).
sample JAVA program (= choose reduced mass or electron mass )
C language program (= choose reduced mass or electron mass )
↑ When you choose the condition of "1 = normal" in the above program, this program calculates the normal helium's energy (= -79.0037 eV ) using the reduced mass except when the center of two electrons is at the nucleus in the rotating helium, which agrees with the experimental helium energy the most perfectly.
Using only reduced mass also agrees with experiments
Even if we use only reduced mass also when a helium nucleus tranisently stops, this Bohr's helium energy (= -79.026 eV ) agrees with experiments (= -79.005 eV ).
| Helium energy (eV) | r1 (MM) | 1/4-orbit wavelength | one-orbit wavelength |
|---|---|---|---|
| -79.0037 | 3075.0 | 0.250037 | 1.000148 |
| -79.0200 | 3074.5 | 0.250011 | 1.000044 |
| -79.0268 | 3074.0 | 0.250000 | 1.000000 |
| -79.0300 | 3074.0 | 0.249995 | 0.999980 |
| -79.0500 | 3073.0 | 0.24996 | 0.999856 |
Always reduced mass condition also agrees with experimental helium.
If you choose "3 = reduced mass condition"
in this or
this program, this program calculates helium energy using only reduced mass in all calculations, which also gives the right energy (= -79.0268 eV in the above table.4 ) agreeing with the experimental helium energy of -79.005 eV.
As a result, this real Bohr's helium model with two perpendicular orbits agrees with experimental helium energy, whether reduced mass or original electron mass is used (= the result of reduced mass is slightly better ).

Feel free to link to this site.