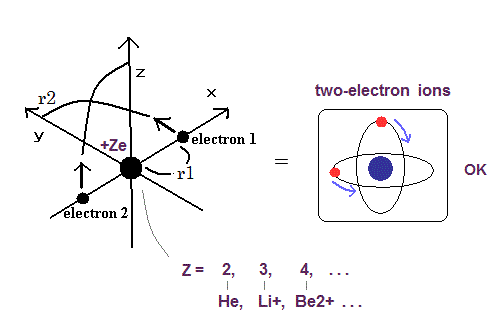
Top page (correct Bohr model including the two-electron atoms).
Electron spin is an illusion !
Our new Bohr model has succeeded in calculating the Helium ionization energy more correctly than the quantum mechanical variational methods as shown in the Top page.
In this new successful Bohr model, the two electrons of the helium atom (He) are moving on the orbits of just one de Broglie's wavelength which are perpendicular to each other.
This model can explain the phenomena of Pauli exclusion principle correctly, because there is no space for the third electron to enter this new two-electron Bohr's 1s state.
Surprisingly, this new atomic structure of the Bohr's helium can be applied to other two-electron atoms (ions ), Lithium ion (Li+), Beryllium (Be2+), Boron (B3+) and Carbon (C4+) ions, too.
(See this page.)
Furthermore, the ionization (ground state) energy of the three-electron atom lithium (Li) can be calculated correctly using the approximate "2S" Bohr orbit. (See this page.)
Fig. 1. Two-electron Atomic Model ( He, Li+, Be2+, B3+, C4+ .. O6+... )

Here we show the ionization energies of oxygen.
| Name | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
|---|---|---|---|---|---|---|---|---|
| Oxygen (O) | 13.618 | 35.117 | 54.935 | 77.413 | 113.899 | 138.119 | 739.290 | 871.410 |
As shown in this Table 1, the ionization energies of 1S electrons (red values) are much bigger than other ionization energies.
This means that also in oxygen atom, only two "1S" electrons are much closer to the nucleus than other electrons.
The sum of the ionization energies of oxygen ion ( O6+ ) is 739.290 + 871.410 = 1610.70 eV
(See the above Table 1. So the experimental value of the total (ground state) energy is -1610.70 eV )
We suppose the two electrons of O6+ are orbiting like Fig. 1.
See the Top page (He) or Li+ page about the detailed computing methods.
Here we use the next sample JAVA program.
(In this program, we use the new units, 1 MM = 10-14 meter, 1 SS = 10-22 second, 1 MM/SS = 108 m/s. )
Sample JAVA program to compute the orbits of the two-electron atoms.
Table 2 shows the results of r1 and WN (number of de Broglie's waves contained in one quarter of the orbit) in which last VY ( y component of electron 1's velocity after moving one quarter of its orbit ) is zero at various energy levels of O6+.
(When the last VY is zero, the electrons move on the stable orbits.)
| E (eV) | r1 (MM) | WN | WN x 4 |
|---|---|---|---|
| -1600.0 | 692.0 | 0.25124 | 1.00496 |
| -1605.0 | 690.0 | 0.25087 | 1.00348 |
| -1610.0 | 688.0 | 0.25045 | 1.00180 |
| -1616.0 | 685.5 | 0.25000 | 1.00000 |
| -1620.0 | 683.5 | 0.24970 | 0.99880 |
| -1625.0 | 681.5 | 0.24932 | 0.99728 |
| -1630.0 | 679.5 | 0.24894 | 0.99576 |
So when the total energy of O6+ is -1616.0 eV, the number of de Broglie's waves contained in one orbits becomes just 1.00000.
As I said above, the experimental value of the O6+ energy is -1610.70 eV.
The error is only 5.3 eV. The ionization energy of O6+ is very big, so the error is only 0.33%.
So it can be said that also in the oxigen ion (O6+), the orbital planes of the two electrons are almost perpendicular to each other.
To confirm the third electron of oxygen enters the orbit of two (not one) de Broglie's wavelength, we try computing the energy of the three-electron oxigen ion (O5+).
Oxygen ion (O5+) has two electrons in 1S orbital, and one electron in 2S orbital.
The two electrons of 1S state are attracted to the 8e+ nucleus strongly, so they are much closer to the nucleus than the 2S electron.
So approximately, we can suppose the 2S electron is moving around the 6e+ nucleus ( +8e - 2e = +6e ) on the circular orbit of the two de Broglie's wavelength.
(This means that we can use the equations of the hydrogen-like atoms in 2S electron approximately.)
According to the Bohr hydrogen-like model, the radius (Rb) of the n × de Broglie's wavelength orbit is,
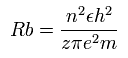
(when z = 1 and n = 1, Rb becomes "Bohr radius".)
In the 2S electron of O5+, the positive charge of the nucleus (z) is supposed to be 6+ (see above), and n is 2.
So this Rb is 4/6 × Bohr radius ( = 0.3527 × 10-10 meter = 3527 MM ) .
(Here we use the new units, 1 MM = 10-14 meter, 1 SS = 10-22 second, 1 MM/SS = 108 m/s )
The energy levels of the hydrogen-like atom is known to be,
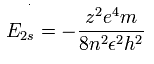
In the 2S electron of O5+, we substitute n=2 and z=6 into this equation.
So we suppose the oxygen ion (O5+) model as follows,
Fig. 2. Oxygen ion (O5+) model
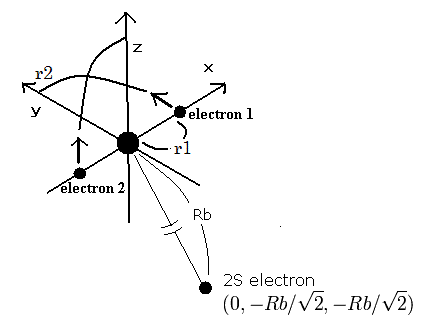
The two electrons of 1S state are avoiding 2S electron, so we fix the 2S electron at ( 0, - Rb / √2, - Rb / √2 ) as shown in Fig. 2.
(Fig. 2. shows the relative positions, But actually, this 2S electron is rotating around the nucleus slower than the 1S electrons.)
Like O6+ (see also top page ), when the electron 1 is at (x, y, 0), the electron 2 is at ( -x, 0, y ).
So the distances among the particles are,
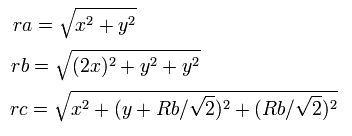
where ra is the distance between the electron 1 and the nucleus, rb is between the two 1S electrons, and rc is between the electron 1 and 2S electron, respectively.
The x component of the acceleration (m/sec2) of the electron 1 is,
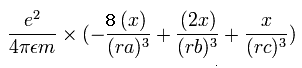
In the same way, the y component of the acceleration (m/sec2) of the electron 1 is,
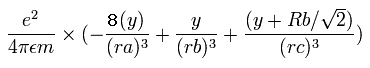
Then we compute the number of de Broglie's waves contained in one quarter of the 1S orbit like O6+
Sample JAVA program (O5+ ion)
Table. 3 shows the results in which the last VY ( y component of electron 1's velocity after moving one quarter of its orbit ) is the closest to zero in O5+.
| E (eV) | r1 (MM) | WN | WN x 4 |
|---|---|---|---|
| -1730.0 | 694.5 | 0.25124 | 1.00496 |
| -1735.0 | 692.5 | 0.25083 | 1.00332 |
| -1740.0 | 690.0 | 0.25046 | 1.00184 |
| -1746.0 | 688.0 | 0.25000 | 1.00000 |
| -1750.0 | 686.0 | 0.24967 | 0.99868 |
| -1755.0 | 684.0 | 0.24930 | 0.99720 |
| -1760.0 | 681.5 | 0.24892 | 0.99568 |
This results shows that when the ground state energy of O5+ ion is -1746.0 eV, the orbital length becomes just 1.00000 × de Broglie's wavelength.
The experimental value of the ground state energy of the oxygen (O5+) ion is = -138.119 -739.290 -871.410 = -1748.8 eV.
So the calculation error is only 2.8 eV ( 0.16 % ).
(This small error is probably caused by using the "approximate" orbit of the 2S electron.)
This means that it is almost correct to say that the three electrons of the oxygen ion (O5+) are moving like Fig.2.
(The third electron of oxygen enters the orbit of 2 ( not 1 ) × de Broglie's wavelength.)
In the neutral oxygen atom (O), how are six outer shell (valence) electrons moving ?
The neutral oxygen has eight electrons, in which two 1S electrons are very close to the nucleus (see above O6+ section).
So approximately we suppose these six electrons are moving around one +6e central nucleus (+8e-2e = +6e).
And six outer shell electrons are thought to be avoiding each other and moving symmetrically around this nucleus.
In the regular polyhedrons, the octahedron has six vertices (= six outer shell electrons ), so first we suppose the six outer shell electrons of oxygen are arranged like octahedron.
(Here, the total energy of the six valence electrons are supposed to be -433.103 eV = sum of 1-6 ionization energies.)
Fig. 3-1. Estimated Oxygen atom. (Octahedron) Nucleus = (0,0,0)
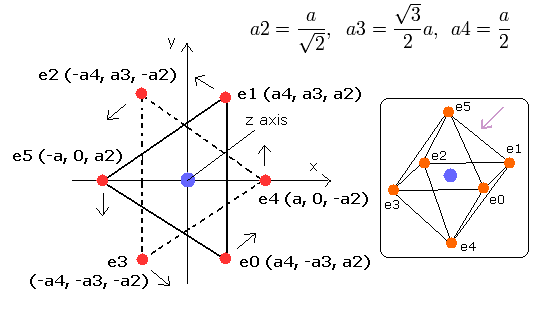
Here, we use the next sample JAVA (version 1.5 (or 5.0)) program to visualize the six electrons' motions.
Sample JAVA program (O visualization)
This program is a little long. So if you copy and paste the above program source code into some text editor,
you can easily compile and run this.
In this program, class file name is oxygen2, so save this text editor as "oxygen2.java", and compile it.
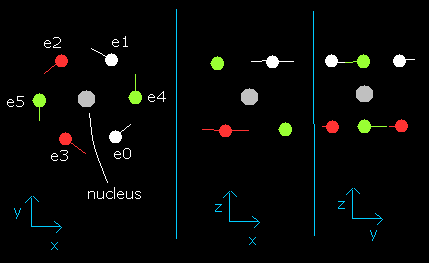
In this program, the oxygen nucleus is shown as a gray circle. (See Fig.3-2.)
Six electrons are white (e0, e1), red (e2, e3), and green (e4, e5) circles.
The x, y and z axes are the same as Fig. 3-1.
The left, center and right figures are x-y, x-z, and y-z planes, respectively.
And the velocity vector of each eletron is shown as a line.
Fig. 3-2. O nucleus and six valence electrons in the program (wn=0)
Each coordinate of electrons (+X (MM), +Y (MM), +Z (MM)) in the text box means "relative" position from the nucleus.
Here we use the new units, as follows.
"nuc (MM)" means the distance between the nucleus and each electron.
"VX: VY: VZ" means the ratio of each component of the initial electron's velocity vector.
So, (VX: VY; VZ)=(100:100:0) is the same as (VX: VY: VZ)=(10:10:0).
When you choose the number (=wn) of de Broglie's waves (0.1 -- 2.0) in the scrollbar, and put "orbit" button, this program starts to compute the electron's orbit until de Broglie's waves contained in the orbit becomes wn.
About the detailed method, see Top page.
At short time intervals (1 SS), we calculate the Coulomb forces among electrons and nuclei, and change each velocity vector based on that result.
And we also compute the number of de Broglie's waves contained in the short segments at each electron.
(In this program, the total energy of the six valence electrons are supposed to be -433.103 eV = sum of 1-6 ionization energy.)
And this program outpus the last coordinate and velocity vector of each electron in both the text box and image area.
When you type the value (=a (MM) of Fig.3-1) in the textbox next to "radiu" button and click this button, the size (radius) of the six electrons' octahedron changes as shown in Fig.3-1.
First, we input the value "3820" as a (MM), and click "radiu" button.
(In this case, the value a of Fig.3-1. becomes 3820 (MM), and the first distance (=nuc) between nucleus and each electron becomes 4678 MM.)
And when we choose the value "0.5" in the scrollbar as the number of de Broglie's waves (wn), and click "orbit" button, the calculation starts and the image result becomes as follows,
Fig. 3-3. Oxygen electrons at wn = 0.5
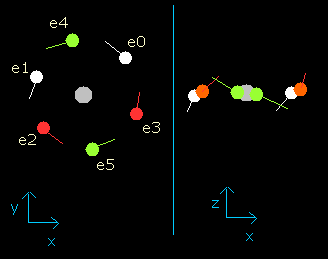
When the electrons have moved their orbits of 0.5 de Broglie's wavelength (wn=0.5), all six valence electrons of oxygen are arranged near the x-y plane (Fig. 3-3, right panel).
And at this point (wn=0.5), the distance between nucleus and electron is about 4204 MM.
Next, click the "retur" button, which go back to the initial condition (wn=0).
And choose the value "0.9" in the scrollbar as the number of de Broglie's waves (wn), and click "orbit" button again. (Fig.3-4)
Fig. 3-4. Oxygen electrons at wn = 0.9
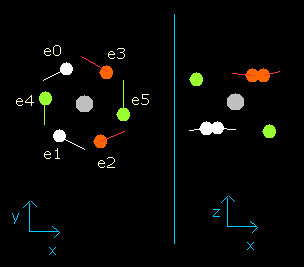
This result shows that when the electrons have moved their orbits of 0.9 (about 1.0) de Broglie's wavelength (wn=0.9), all six valence electrons of oxygen are on the opposite side from their initial positions.
At this point (wn=0.9), the distance between mucleus and electron becomes the shortest. ( = 3801 MM).
Click the "retur" button again, choose the value "1.9", and click "orbit" button.
The result is, (Fig. 3-5)
Fig. 3-5. Oxygen electrons at wn = 1.9 (=almost same as wn=0.0)
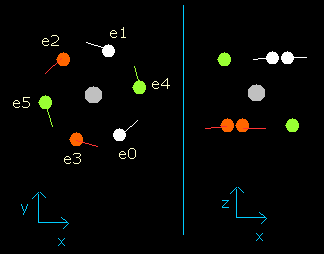
So this result proves that the all six electrons go back to almost original position when the the number of de Broglie's waves contained in their orbits becomes 1.9 (almost 2.0).
(Probably, these orbits are precessing or swaying a little.)
If you set a=3100 MM, which means the initial distance between the nucleus and electrons is 3796 MM, this distance (nuc) becomes the longest at wn=0.9 (4677 MM).
Try this, too.
In the above program. we suppose the "central positive charge" is just +6.0e (= +8e (nucleus) -2e (two 1s electrons) = +6e)
But as shown in the above section, the two 1S electrons are about 700 MM apart from +8e oxygen nucleus. (They don't stick to the nucleus.)
So, the "effective" positive charge which influences the six valence electrons are probably a little bigger than +6.0e.
Here, we try this orbital calculation again, supposing this central positive charge is +6.3e instead of +6.0e
Sample JAVA program. (central charge +6.3e instead of +6.0e)
This sample program is almost same as the above sample program except for the central positive charge (+6.0e --> +6.3e).
In this program, when the orbital length becomes just 2.0 de Broglie's wavelength, all six valence electrons of oxygen can return to their original positions, as follows,
Fig. 3-6. Oxygen electrons at wn = 0.0 and wn = 2.0 ("effective" charge = +6.3e)
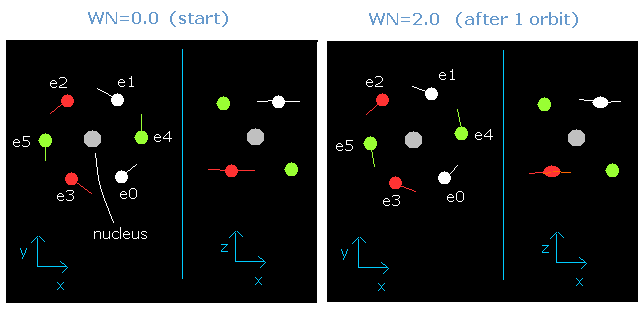
This result is very important.
When the total energy of the six valence electrons is determined (=-433.103 eV), the average length (or radius) of one-round orbit is fixed.
(This means that if the total energy of electrons is determined, the average length and the number of de Broglie's waves contained in the one-round of the orbit is fixed "automatically".)
And this number proves to be 2.0 in the case of neutral oxygen atom.
(So this result shows that the six valence electrons of oxygen atom are moving on the orbits of 2.0 de Broglie's wavelength.)
In the above O5+ ion, we use the +6.0e central charge, because at the starting point of Fig.2, the error by this approximate methods is very small.
See also this page (the relation between inner 1S and 2S electrons).
When the central positive charge (Z) is just +6.0e (=+8e-2e), the number of de Broglie's waves (wn) contained in one orbit is 1.9.
But actually the two 1S electrons don't stick to the +8e nucleus, so if we suppose this charge (Z) is +6.3e (a little bigger than +6.0e), the de Broglie's wave becomes just 2.0, as shown above.
Here we try to prove this using the Virial theorem.
We suppose that the six valence electrons are arranged "octahedrally" around +Ze central charge, as follows,
Fig. 4. Oxygen valence electrons ("effective" charge = +Ze)
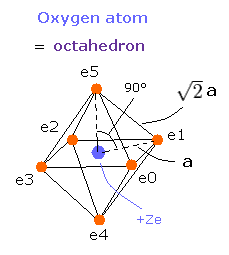
In Fig. 4, the distance between an electron and the central positive charge is a.
According to the Virial theorem, "average" total potential (V) and kinetic (T) energies satisfy the relation,
< V > = -2 × < T > , E = V + T
The total energy (E) of the six valence electrons is -433.103 eV, so this potential energy (V) becomes 2 × -433.103 = -866.206 eV.
When we calculate this potential energy (V) using Fig. 4 model, this V ( eV --> J ) becomes,
(by calculating the interactions between electron - nucleus and electron-electron,)
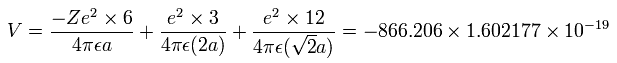
And the total kinetic energy (T) of the six electrons is 433.103 (eV), as follows,
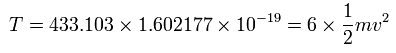
And by calculating the Coulomb forces among the particles of Fig.4, the force (F) acting on electron 0 (toward nucleus) becomes,
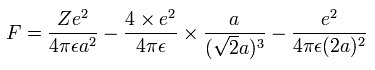
Using the above velocity (v) (of T) and F, we suppose the centrifugal force is equal to the force (F), as follows,
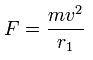
This r1 is "temporary" radius, which appears by this relation.
Using this r1, we can calculate the number of de Broglie's waves (wn) contained in one orbit, as follows,
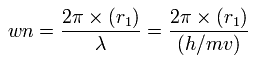
When Z = 6.0, this wn becomes 1.88 (almost 1.9).
And when Z = 6.3, this wn becomes just 2.01.
So we can prove this fact using the Virial theorem.
As shown in the O6+ and O5+ ion sections, the distance between two 1S electrons and nucleus is about 700 MM.
So this result (in which the shortest distance is about 3800 MM ) means that six valence electrons don't collide with the inner 1S electrons, and can move safely.
For example, the O-H bond length of water (H2O) is 9584 MM, so this computing value 4678 MM is thought to be proper.
By the way, the six outer shell electrons of oxygen atom are really arranged octahedorally ?
In the case of carbon, the four outer shell electrons of carbon atom are arranged tetrahedrally. (See also Bohr model carbon page).
In the regular tetrahedron, the angle between the segments joining the center (nucleus) and the vertices (electrons) is 109.5 degrees.
Actually, in the methane (CH4), four electrons are arranged tetrahedrally, and the H-C-H angle is just 109.5 degrees.
Also in the carbon-like atom silicon (Si), a silicon-hydrogen compound, monosilane (SiH4) has tetrahedral structure, too. (H-Si-H angle is 109.5 degrees).
In the water (H2O), two hydrogen atoms and two lone pairs of oxygen's electrons are said to be arranged "tetrahedrally".
But the H-O-H angle is 104.45 degrees, which is a little smaller than 109.5 degrees.
Why is this angle smaller than 109.5 degrees ?
In the regular octahedron, the angle between the segments joining the center (nucleus) and the vertices (electrons) is 90 degrees.
So 104.45 is between 90 and 109.5 degrees.
If the atomic structure of the oxygen atom is octahedron, why isn't H-O-H angle 90 degrees ?
Surprisingly, in the other oxygen-like atoms, sulfur (S), Selenium (Se), and Tellurium (Te), which have six valence electrons, H-S-H, H-Se-H, and H-Te-H angles are 92.1, 91.0, and 89.5 degrees, respectively. ( = almost 90 degrees).
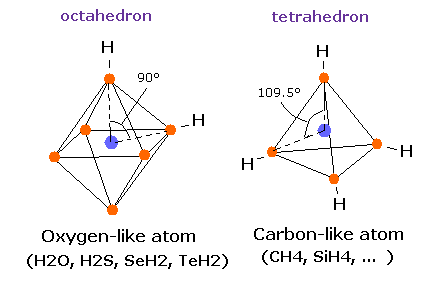
So in these oxygen-like atoms, the six valence electrons can be said to be arranged almost "octahedrally".
In the case of oxygen, the hydrogen atom is closer to the oxygen atom than other oxygen-like atoms.
So, it is thought that due to the repulsive Coulomb forces among oxygen's and hydrogen's electrons (or between two hydrogen's nuclei), water (H2O) can not keep "octahedral" structure.
(If the two hydrogen atoms are just opposite sides of the oxygen atom, electrons are attracted to the two hydrogen nuclei in the opposite directions.
So the electron's density around the oxygen nucleus becomes "unnaturally" low, which case probably won't happen.)

2010/8/18 updated. Feel free to link to this site.